Moist Air
So far we only considered dry air, but already learned that moisture affects pressure, the molecular weight of the air parcel and hence its individual gas constant.
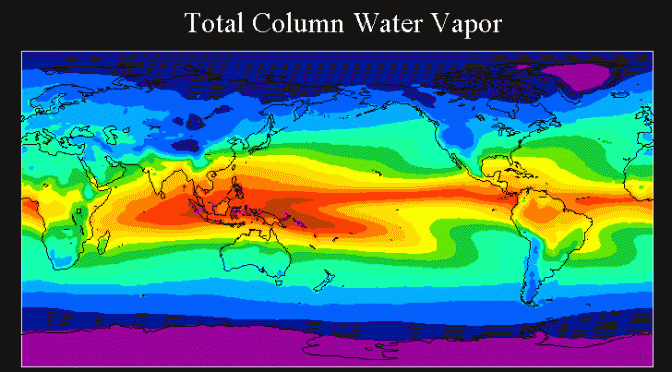
Moist air is important for weather, climate, air quality despite its low, strongly varying fraction of 0 to 4% as we had discussed in Unit 1. Besides its role in the water cycle, water vapor is also playing a large role in the energy-, and trace gas cycle. Water vapor is involved in cloud and precipitation formation, affects radiation, and many gas-phase chemical and aerosol processes.
Water is the only atmospheric constituent that occurs in all its phases in the troposphere. For simplicity we will deal with the moist atmosphere first before, in a later unit, we address phase transition processes between gaseous, liquid & solid states of water. The moisture measures we define in this unit only apply to water vapor.
Learning goal
Describing the moist atmosphere quantitatively and qualitatively.
After completion of this unit students will be able to
- Define the various moisture measures
- Explain which moisture measures are conserved when an air parcel moves vertically and why
- Express atmospheric moisture in various measures
- Proof whether or not a moisture measure is conservative (graduate students)
Students’ Tasks
- Watch the video
- Read chapter 2.7 to 2.7.6 (included) of Lectures in Meteorology
- ATM401 students: Answer the questionnaire by Thursday 2359 AST . ATM601, CHEM601 students: Sent your summary of the material by Thursday 2359 AST .
- All students: Participate in the discussion channel of this unit on the discussion board
- All students: Solve the problems listed in this unit 5 application sheet at your class level and submit the scanned solutions to cmoelders@alaska.edu.
Supplemental material
In case you want to take notes on the slides used in the video, here they are.
Worked example problem
Show that water vapor pressure decreases when the air parcel expands under isotherm conditions.
Further reading
Lecture on how to measure atmospheric humidity
© 2019-2020 Nicole Mölders | All rights reserved